Eleftha 440mg Multi Dose Vial
Price 1250 INR/ Unit
Eleftha 440mg Multi Dose Vial Specification
- Salt Composition
- Trastuzumab (440mg)
- Dosage Form
- Injection
- Indication
- Breast cancer, Gastric cancer
- Feature
- Multi-dose vial for intravenous use
- Ingredients
- Trastuzumab Lyophilized Powder, Excipients
- Application
- Hospital, Oncology Use
- Ph Level
- 6.0 (approx.)
- Physical Color/Texture
- White to pale yellow lyophilized powder
- Fermentation Smell
- Odorless
- Storage Instructions
- Refrigerate at 2C - 8C. Do not freeze.
- Shelf Life
- 24 months
- Sterility
- Sterile powder
- MRP Visibility
- On unit box
- Her2 Status Required
- Yes
- Container Type
- Glass vial
- Diluent Included
- Yes (bacteriostatic water)
- Prescription Required
- Yes
- Product Code
- 440mg vial
- Reconstitution Required
- Yes
- Administration Route
- IV infusion
- Pack Size
- 1 vial per box
- Batch Number Format
- Printed on pack
Eleftha 440mg Multi Dose Vial Trade Information
- Minimum Order Quantity
- 1 Unit
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 1000 Units Per Month
- Delivery Time
- 7 Days
- Main Domestic Market
- All India
About Eleftha 440mg Multi Dose Vial
Diverse Clinical Applications and Specific Oncology Use
Eleftha 440mg Multi Dose Vial is primarily indicated for HER2-positive breast cancer and gastric cancer, offering reliable therapy within hospital oncology protocols. Its specific usage requires the reconstitution of sterile, lyophilized powder in bacteriostatic water for IV infusion. Application media involves administration via healthcare professionals, ensuring optimal safety and efficacy for each patient. Eleftha's multi-dose nature enhances hospital inventory management by supporting multiple infusions from a single vial, thereby reducing waste and improving workflow.
Export Markets, Sample Policy, and Secure Packaging Exchange
Eleftha 440mg Multi Dose Vial is actively exchanged in major export markets, facilitated by robust order processing systems for hospital and clinical buyers. Sample policies are available upon request, enabling healthcare providers to evaluate the product before large-scale procurement. Packaging details include secure glass vials within unit boxes, marked with batch numbers for traceability, ensuring safe goods transport. Our streamlined approach to packaging and logistics guarantees product integrity and timely delivery from India to global destinations.
FAQ's of Eleftha 440mg Multi Dose Vial:
Q: How is Eleftha 440mg Multi Dose Vial administered?
A: Eleftha 440mg is administered as an intravenous (IV) infusion after reconstituting the sterile lyophilized powder with the included bacteriostatic water, under the supervision of qualified healthcare professionals.Q: What are the specific storage requirements for this vial?
A: The vial must be refrigerated at 2C to 8C and should not be frozen. This helps maintain the sterility and efficacy of the product until its 24-month expiry date.Q: When should Eleftha 440mg be considered for treatment?
A: It is prescribed for patients diagnosed with HER2-positive breast cancer or gastric cancer. The decision to use Eleftha should be made by an oncologist based on HER2 test results.Q: Where is Eleftha 440mg exported from and who are the suppliers?
A: Eleftha 440mg Multi Dose Vial is exported from India by reliable exporters, traders, and suppliers specializing in hospital oncology products.Q: What benefits does the multi-dose vial format offer?
A: The multi-dose format allows multiple infusions from a single vial, improving hospital workflow, reducing medication waste, and enhancing overall cost-effectiveness for healthcare facilities.Q: What quality assurance features does Eleftha 440mg provide?
A: Each unit is batch numbered and MRP-marked on its packaging for traceability and transparency, ensuring that healthcare buyers can verify product authenticity and compliance.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Anti Cancer Injection Category
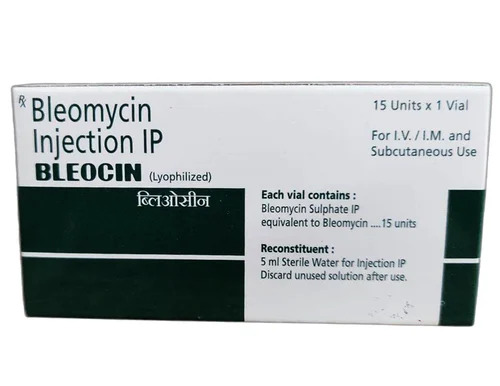
Bleocin Bleomycin Injection IP
Price 480 INR / Vial
Minimum Order Quantity : 20 Vials
Storage Instructions : Store in a refrigerator (2 8C). Do not freeze.
Feature : Other, Vial for injection
Physical Color/Texture : Other , White lyophilized powder
Application : Other, Clinical
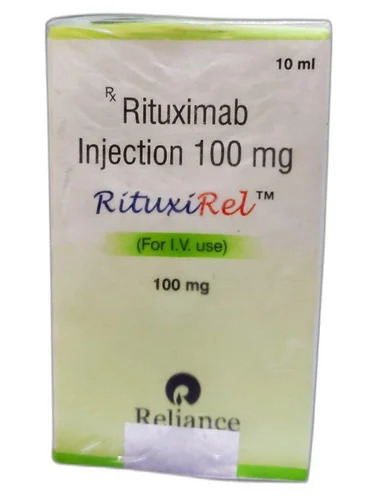
Rituxirel 100mg Rituximab Injection
Price 2200 INR / Vial
Minimum Order Quantity : 2 Vials
Storage Instructions : Store at 2C 8C. Do not freeze.
Feature : Other, Prescription only, cytotoxic agent
Physical Color/Texture : Other , Clear, colorless solution
Application : Other, Clinical
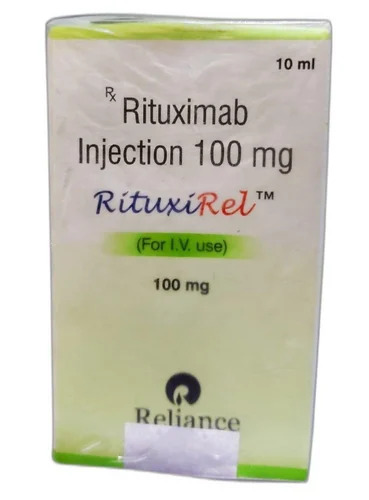
RutuxiRel Rituximab Injections
Price 2200 INR / Vial
Minimum Order Quantity : 1 Vial
Storage Instructions : Store at 2C to 8C
Feature : Other, Lyophilized Powder
Physical Color/Texture : Other , Clear solution
Application : Other, Cancer Treatment
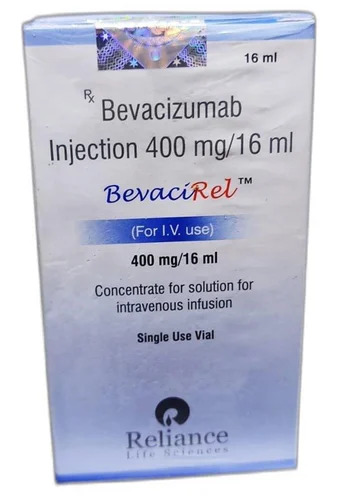
BevaciRel 400mg Bevacizumab Injection
Price 12500 INR / Unit
Minimum Order Quantity : 1 Unit
Storage Instructions : Store in a refrigerator (2 8C). Do not freeze.
Feature : Other, Anticancer monoclonal antibody
Physical Color/Texture : Other , Clear, colorless to pale brownishyellow solution
Application : Other, Clinical
 |
KETHAN PHARMA DISTRIBUTORS
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |






 Send Inquiry
Send Inquiry