|
Z-Taxel 260 Paclitaxel Injection IP
Price 500 INR/ Unit
Z-Taxel 260 Paclitaxel Injection IP Specification
- Indication
- Cancer treatment
- Dosage Form
- Injection
- Salt Composition
- Paclitaxel
- Feature
- Prescription only
- Ingredients
- Paclitaxel IP
- Application
- Intravenous use
- Physical Color/Texture
- Clear, colorless to pale yellow solution
- Fermentation Smell
- Odorless
- Storage Instructions
- Store below 25C, protect from light
- Shelf Life
- 24 Months
- PCD Pharma
- Available
- Container Type
- Vial
- Prescription Required
- Yes
- Sterility
- Sterile solution
- Usage
- Hospital supply
- Packaging Size
- 43.14ml
- Strength
- 260 mg/43.14 ml
Z-Taxel 260 Paclitaxel Injection IP Trade Information
- Minimum Order Quantity
- 20 Units
- Payment Terms
- Cash in Advance (CID)
- Supply Ability
- 1000 Units Per Month
- Delivery Time
- 7 Days
- Main Domestic Market
- All India
About Z-Taxel 260 Paclitaxel Injection IP
Directions and Application Area for Z-Taxel 260 Paclitaxel Injection IP
Z-Taxel 260 is designated exclusively for hospital use, targeting various cancer types through intravenous administration. The surface of application is strictly intravenous-professionals must ensure precise dosage as prescribed. Preparation and infusion should occur under sterile conditions, adhering to professional standards for oncology infusions. Dosage and frequency are tailored as per the physician's discretion, under close clinical supervision, maximizing therapeutic effect and minimizing risk for the patient.
Main Domestic Market, Certification, and Packaging Information
Z-Taxel 260 Paclitaxel Injection IP is primarily available in the eminent Indian market, with robust certifications underscoring its quality assurance. Each arrival of product features radiant packaging in sturdy vials, ensuring sterility and potency are uncompromised. The list price reflects its meritorious efficacy and compliance with clinical safety norms. Advance purchase options and detailed exporter credentials further enhance accessibility for suppliers and traders nationwide.
FAQ's of Z-Taxel 260 Paclitaxel Injection IP:
Q: How should Z-Taxel 260 Paclitaxel Injection IP be administered?
A: This injection is administered intravenously by qualified healthcare professionals in a hospital setting, ensuring precise control over dosage and patient safety.Q: What cancers is Z-Taxel 260 Paclitaxel Injection commonly used to treat?
A: Z-Taxel 260 Paclitaxel Injection is primarily indicated in the treatment of various cancers, including ovarian, breast, and non-small cell lung cancer, among others as directed by the physician.Q: When should Z-Taxel 260 Paclitaxel Injection be purchased and stored?
A: Advance purchase is recommended due to its 24-month shelf life. Store the vials below 25C and protect them from light to preserve quality and potency.Q: Where can Z-Taxel 260 Paclitaxel Injection IP be sourced from?
A: It can be procured from leading exporters, suppliers, traders, and PCD Pharma channels throughout India, with all products provided on prescription basis.Q: What is the process for redeeming the full therapeutic benefit of Z-Taxel 260 Paclitaxel Injection?
A: Regular administration as per the oncologist's guidance and adherence to prescribed dosing schedules ensures patients receive the greatest therapeutic benefit from this medication.Q: What certifications attest to the quality of Z-Taxel 260 Paclitaxel Injection IP?
A: Z-Taxel 260 is supplied with requisite certifications reflecting its safety and efficacy, conforming to industry standards and regulatory guidelines in the domestic market.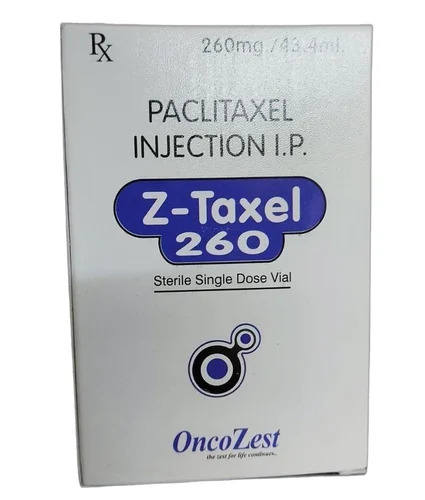
More Products in Anti Cancer Injection Category
Eleftha 440mg Multi Dose Vial
Price 1250 INR / Unit
Minimum Order Quantity : 1 Unit
Ingredients : Other , Trastuzumab Lyophilized Powder, Excipients
Shelf Life : 24 months
Application : Other, Hospital, Oncology Use
Indication : Breast cancer, Gastric cancer
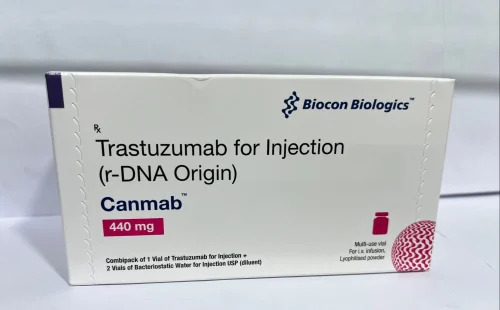
Canmab 440mg Injection
Price 8000 INR / Vial
Minimum Order Quantity : 50 Vials
Ingredients : Other , Trastuzumab
Shelf Life : 24 months
Application : Other, Hospital, Clinical
Indication : HER2positive breast cancer, HER2positive gastric cancer
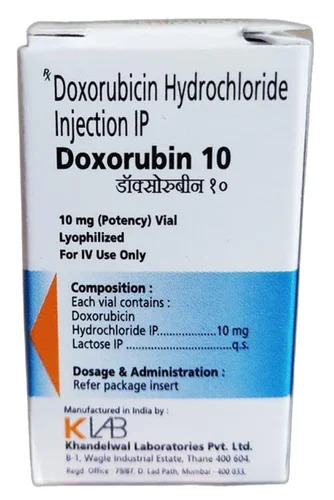
Doxorubin 10 Doxorubicin Hydrochloride Injection IP
Price 80 INR / Unit
Minimum Order Quantity : 10 Units
Ingredients : Other , Doxorubicin Hydrochloride
Shelf Life : 24 Months
Application : Other, Oncology
Indication : Cancer
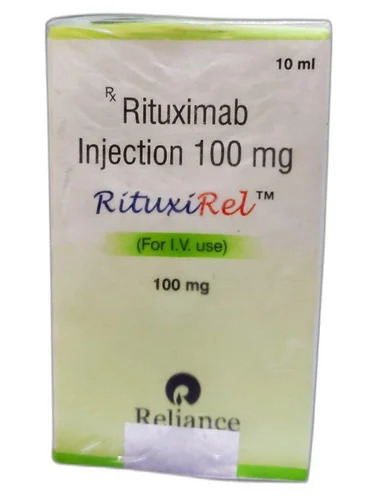
Rituxirel 100mg Rituximab Injection
Price 2200 INR / Vial
Minimum Order Quantity : 2 Vials
Ingredients : Other , Rituximab
Shelf Life : 36 Months
Application : Other, Clinical
Indication : Blood Cancer, Rheumatoid Arthritis
 |
KETHAN PHARMA DISTRIBUTORS
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |







 Send Inquiry
Send Inquiry